 The molar mass of a molecule is equal to the weighted sum of the molar masse of the atoms that compose it. The molar mass of an atom is directly read in the periodic table. This number corresponds to the number of Avogadro `N_A`. The symbol used is M and the usual unit is g/mol (corresponds to kg/mol in the international system).Īs a reminder, a mole of a chemical element is the amount of substance containing `6,02214076*10^23` of elementary entities of that element (atoms, molecules, elemental particle, etc.). The molar mass of an atomic or molecular chemical element is the mass of a mole of that substance.  
If you type SI for Silicon instead of Si, then the calculator will interpret this formula as. Examples: Fe, Co and Xe not FE, CO, XE, fe, ce and xe. Dissolves violently in water with liberation of much heat. The symbol of an atom with two letters has the first letter in uppercase and the second in lowercase. This produces flammable/explosive gas (hydrogen - see ICSC 0001). The symbol of an atom with a single letter is always written in capital letters. So, you need to choose "disable" field 1 to calculate field 2 and disable field 1 and 2 to calculate field 3.ĭo respect the uppercases and lowercases letters when you enter a chemical formula in the calculator above (3rd field). 3rd field: the molar mass of the formula entered (atom or molecule). 2nd field: the molecule mass of the selected molecule (choice from a list of common molecules) 1st field: the molar mass of the selected atom (choice from the 118 elements of the periodic table). CALCIUM CHLORIDE MOLAR MASS HOW TOThis site explains how to find molar mass.The tool calculates in order of priority: 2H 2 O by taking the mass from line 1 of the data table and dividing by the molar mass just found above. In Imperial or US customary measurement system, the density is equal to 114.243 pound per cubic foot lb/ft, or 1.058 ounce per cubic inch oz/inch. density of calcium chloride tetrahydrate is equal to 1 830 kg/m. Calculate the moles of Calcium chloride dihydrate: Cl 2. Calcium chloride tetrahydrate weighs 1.83 gram per cubic centimeter or 1 830 kilogram per cubic meter, i.e. The reason is that the molar mass of the substance affects the conversion. Calculate the molar mass of Calcium chloride dihydrate: CaCl 2. To complete this calculation, you have to know what substance you are trying to convert. Supplier details: American Elements 10884 Weyburn Ave. Relevant identified uses of the substance: Scientific research and development. These relative weights computed from the chemical equation are sometimes called equation weights.Ī common request on this site is to convert grams to moles. Product Number: All applicable American Elements product codes, e.g. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.įinding molar mass starts with units of grams per mole (g/mol). What is the total relative molecular mass of the two moles of calcium chloride Given the atomic masses, Ca. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. Calcium chloride has the molecular formula of CaCl2. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. Molecular weight of Calcium Chloride Molecular Weight Calcium Chloride molecular weight Molar mass of CaCl2 110.984 g/mol Convert grams Calcium Chloride to moles or moles Calcium Chloride to grams Molecular weight calculation: 40.078 + 35. 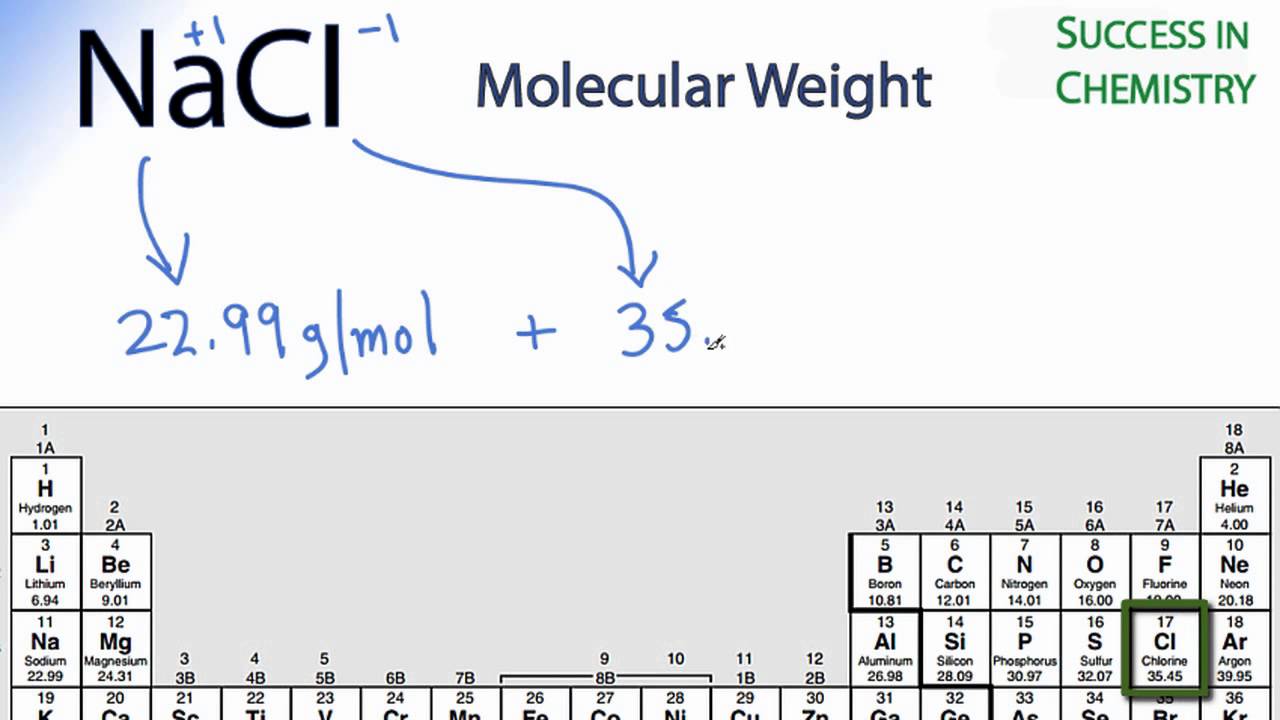 
In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
